Functional groups in organic chemistry Functional groups are structural features distinguish one organic molecule from another. They determine a molecule’s geometry, physical properties, and reactivity. Actually, a functional group is an atom or a group of atoms with characteristic chemical and physical properties. It is the reactive part of the molecule. The functional group is bonded to the carbon backbone or skeleton which consists of carbon-carbon and carbon-hydrogen σ bonds. These bonds are strong, nonpolar, and not readily broken. This carbon skeleton can contain some other atoms of the so-called heteroatoms. This can be nitrogen, oxygen, sulfur, phosphorus and the halogens. Heteroatoms have lone pairs and create electron-deficient sites on carbon. The carbon skeleton can also contain multiple bonds (π bonds). The most common π bonds occur in carbon-carbon and carbon-oxygen double bonds. They are easily broken in chemical reactions and makes a molecule a base or a nucleophile.
Broadway & 50th St 🏙️: BROADWAY 🗽: The Birth of Venus (Bouguereau): BROADWAY 🗽: The Birth of Venus (Bouguereau) : The Birth of Venus (French: La Naissance de Vénus) is one of the most famous paintings by 19t...

















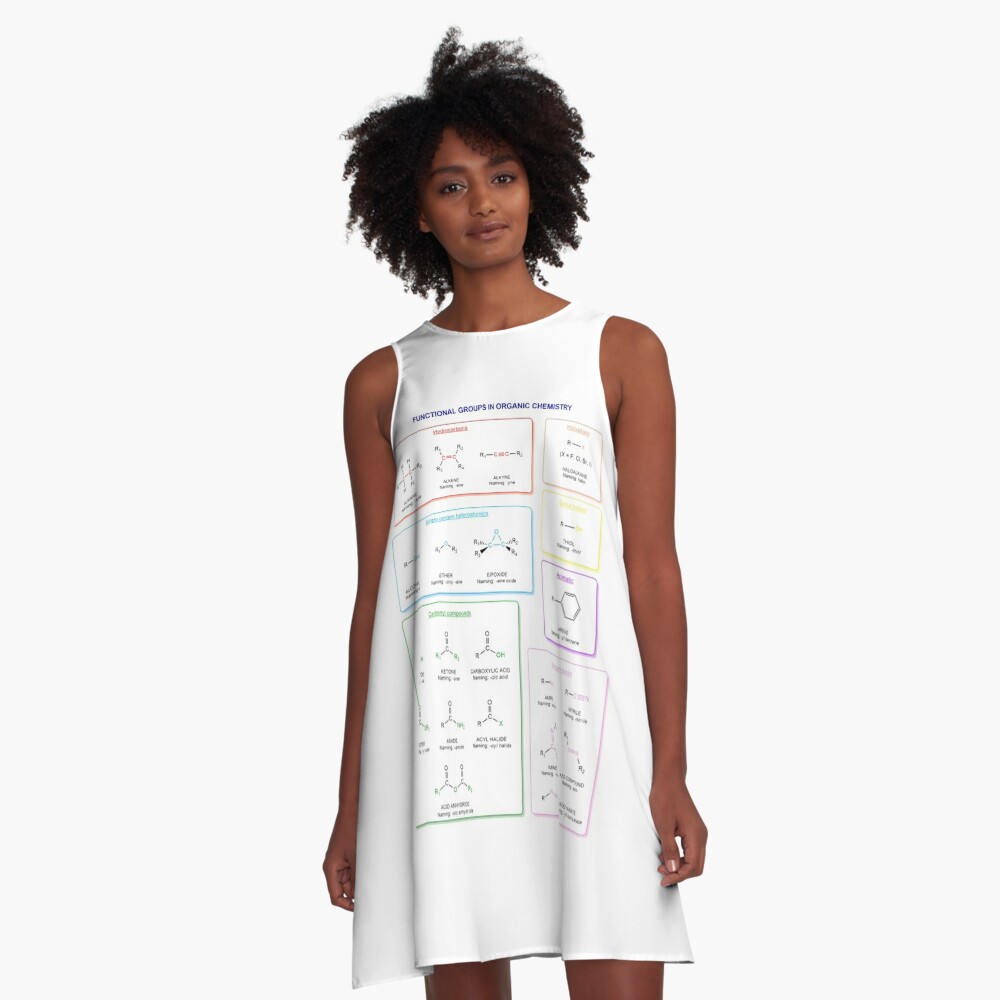






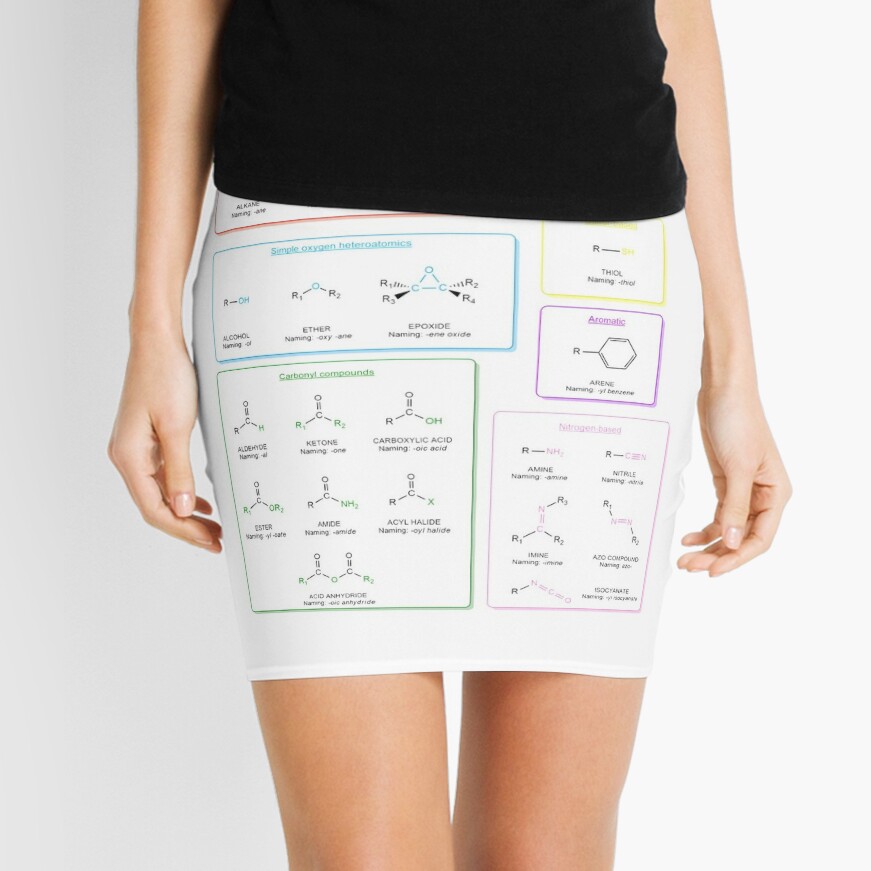


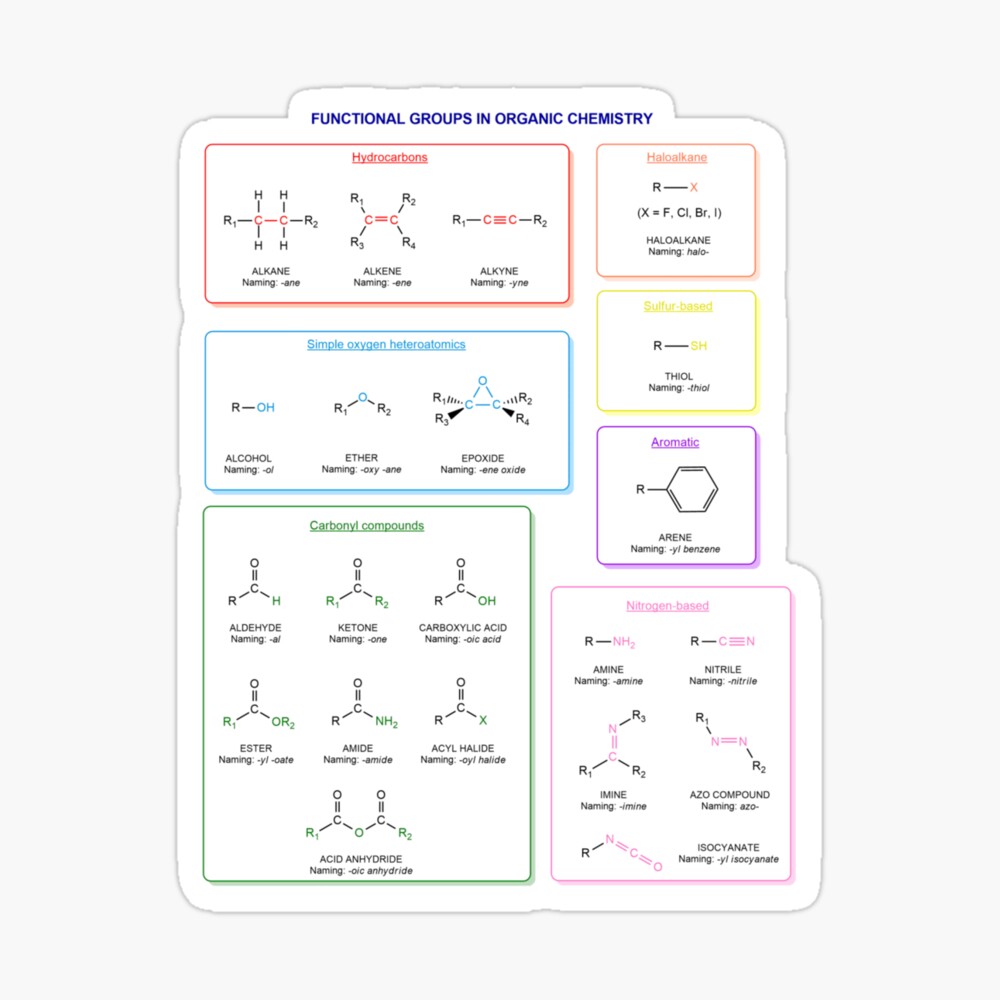
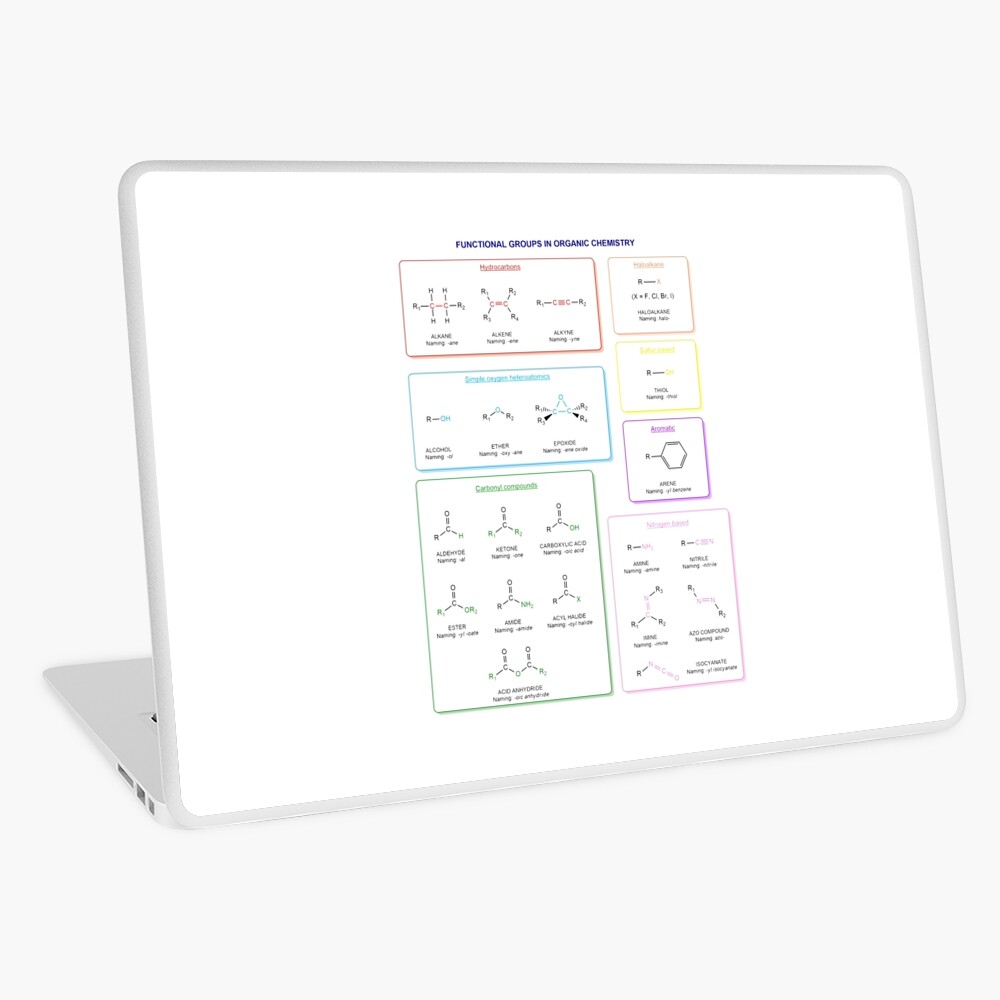
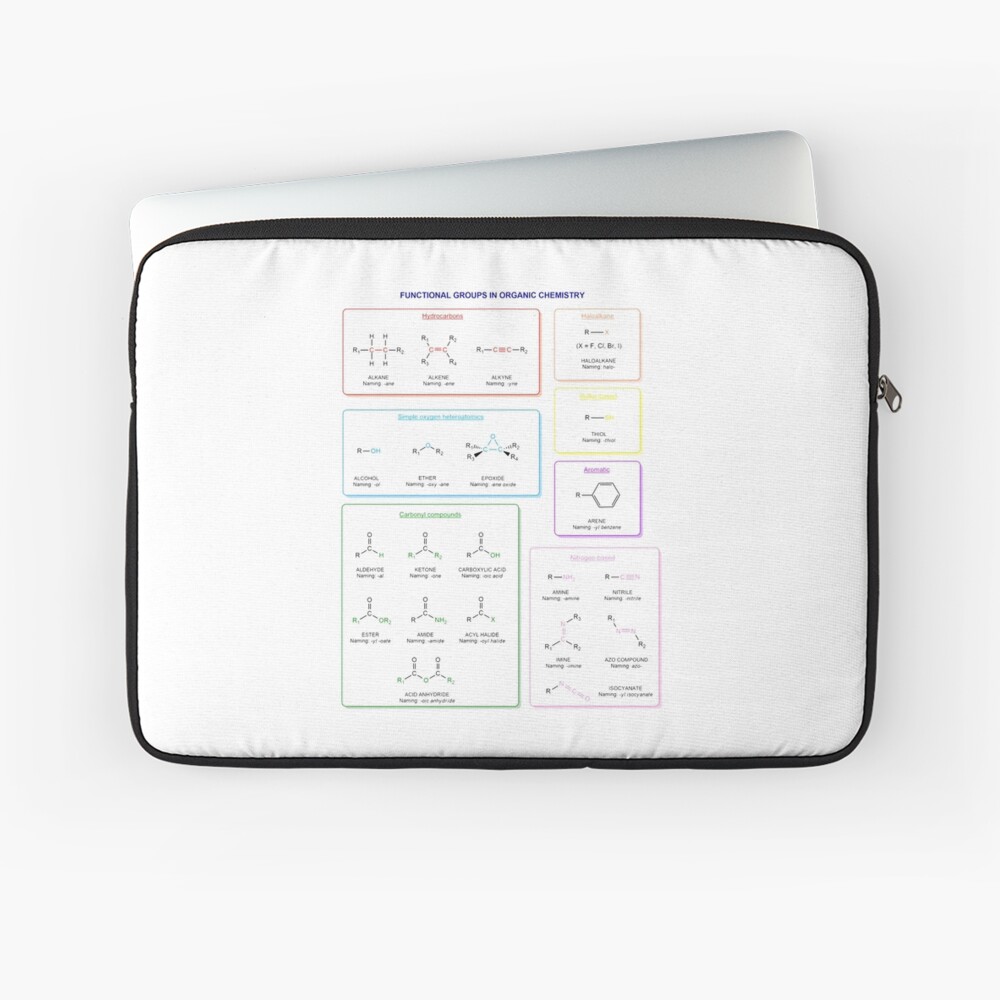
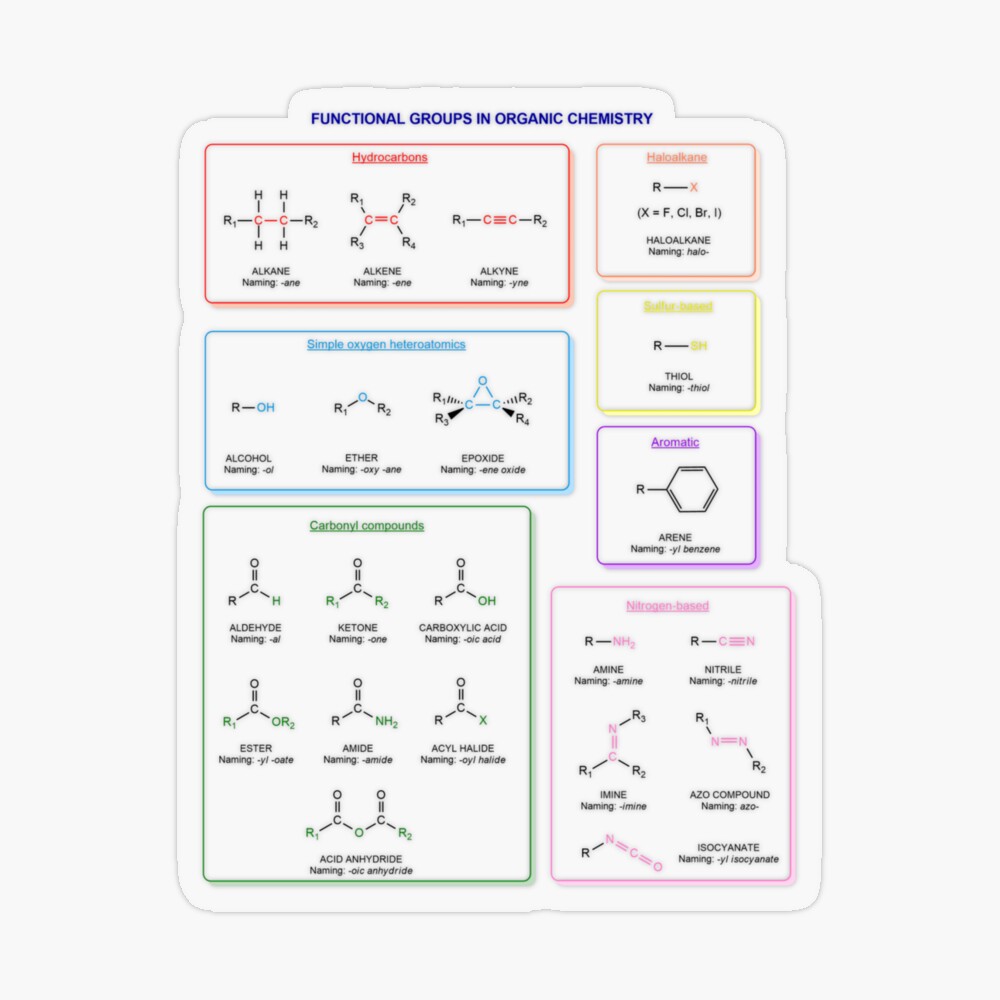

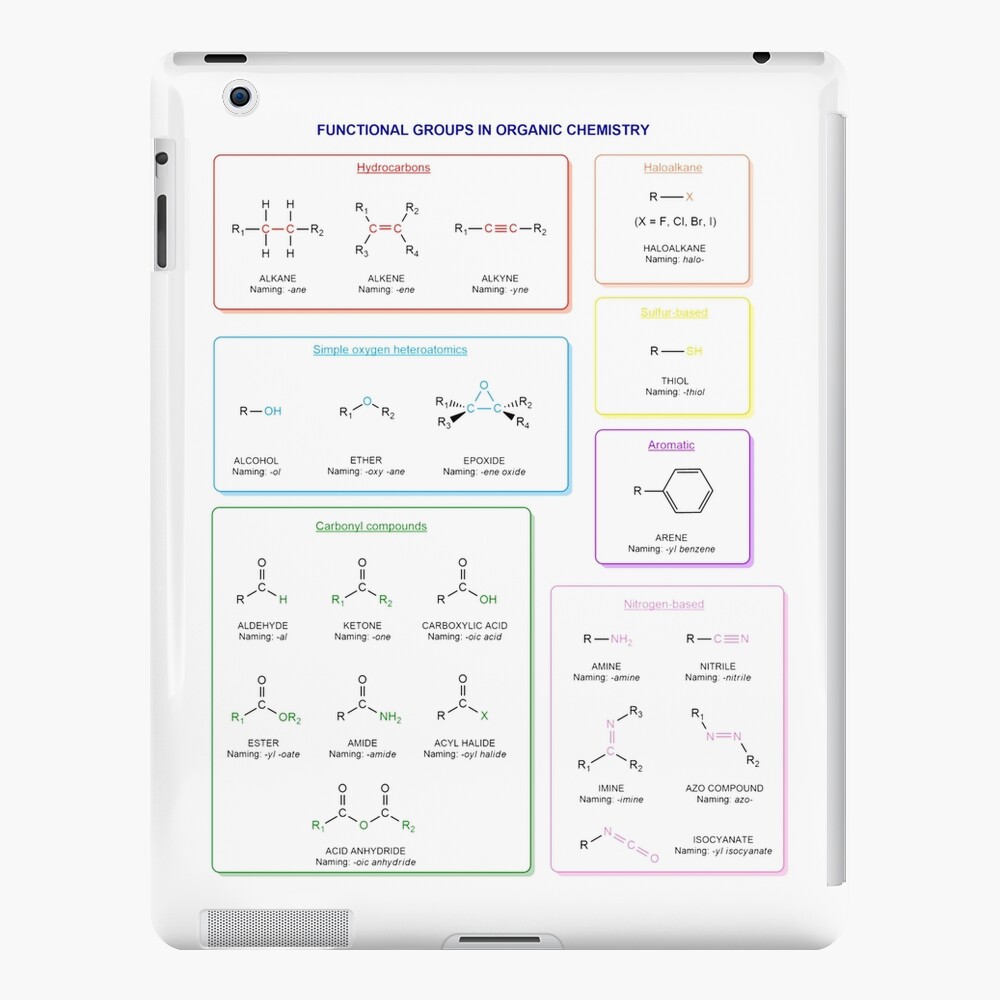





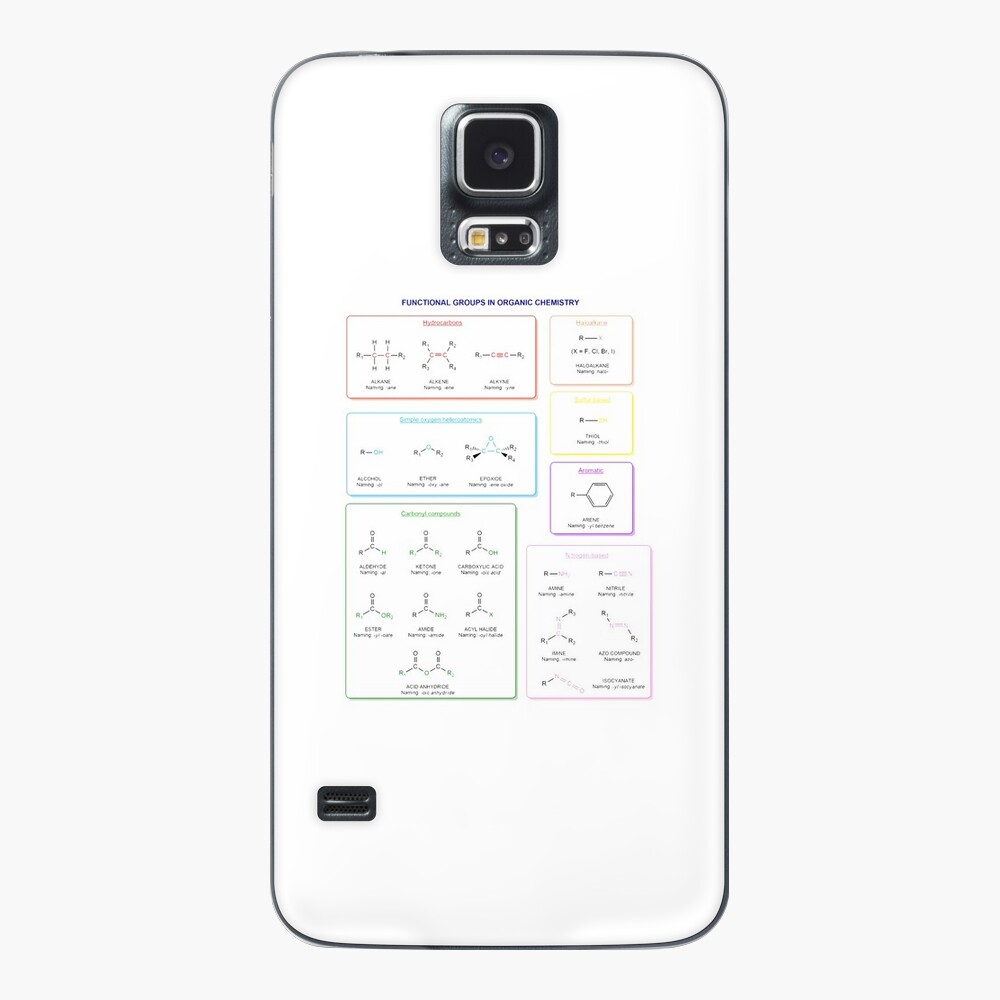
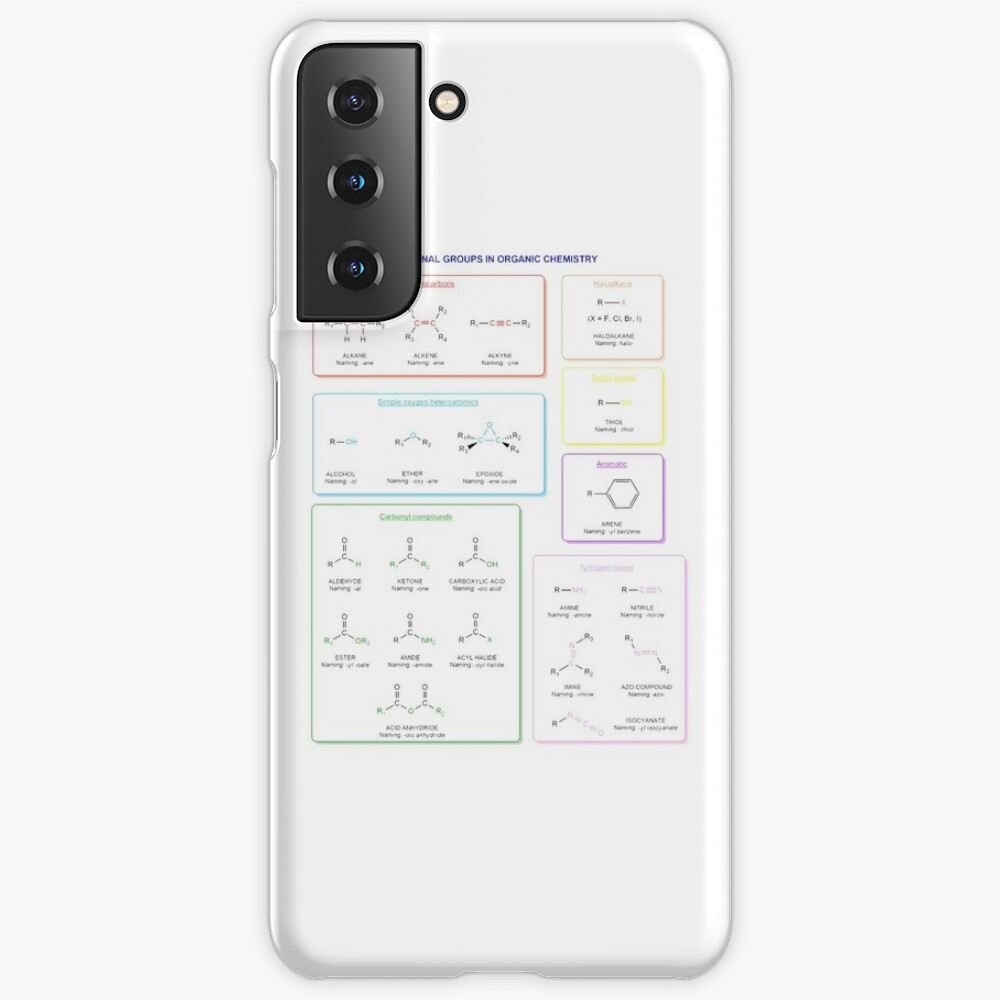

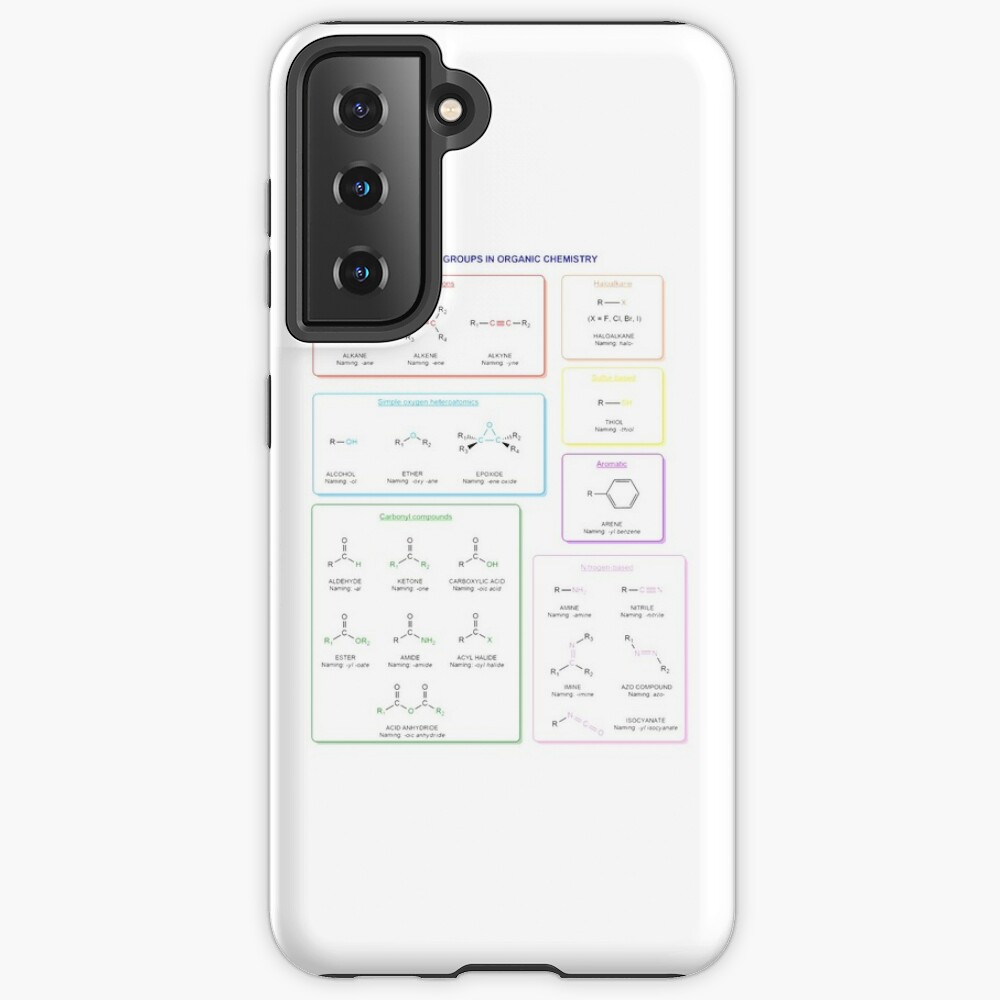

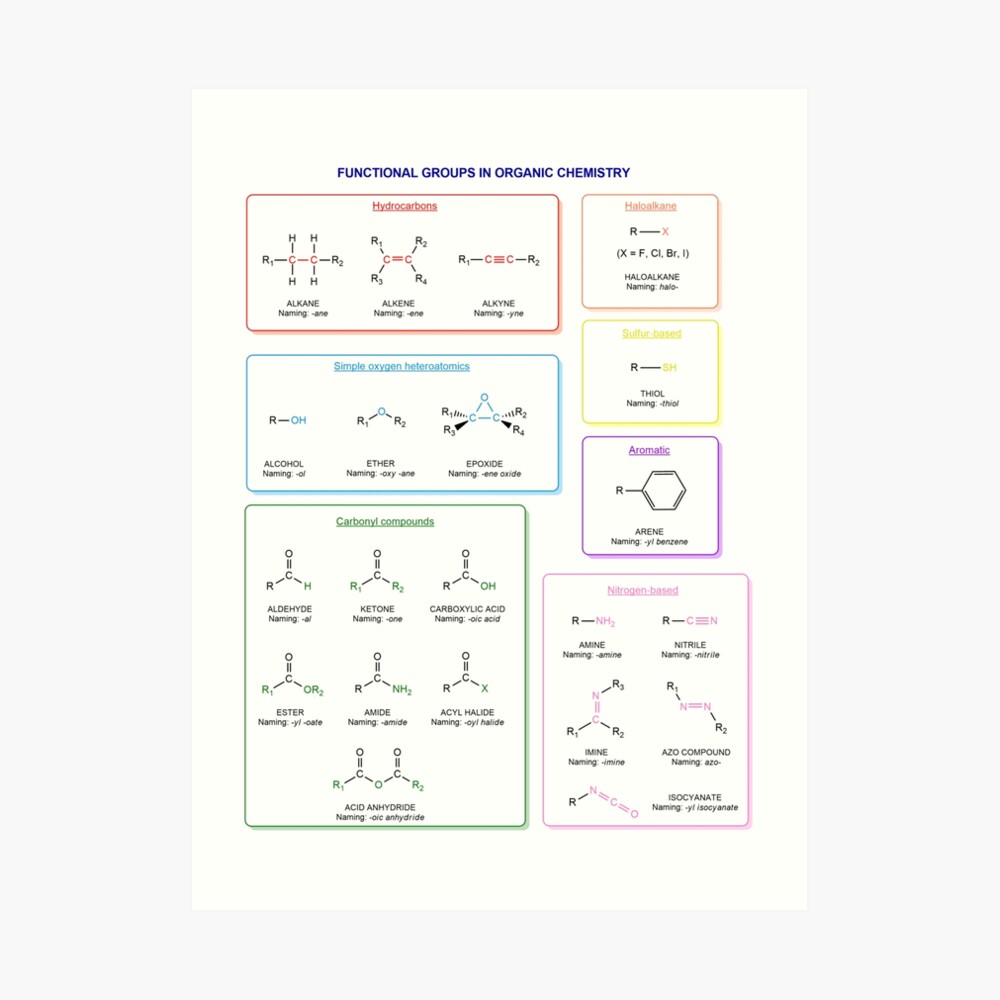

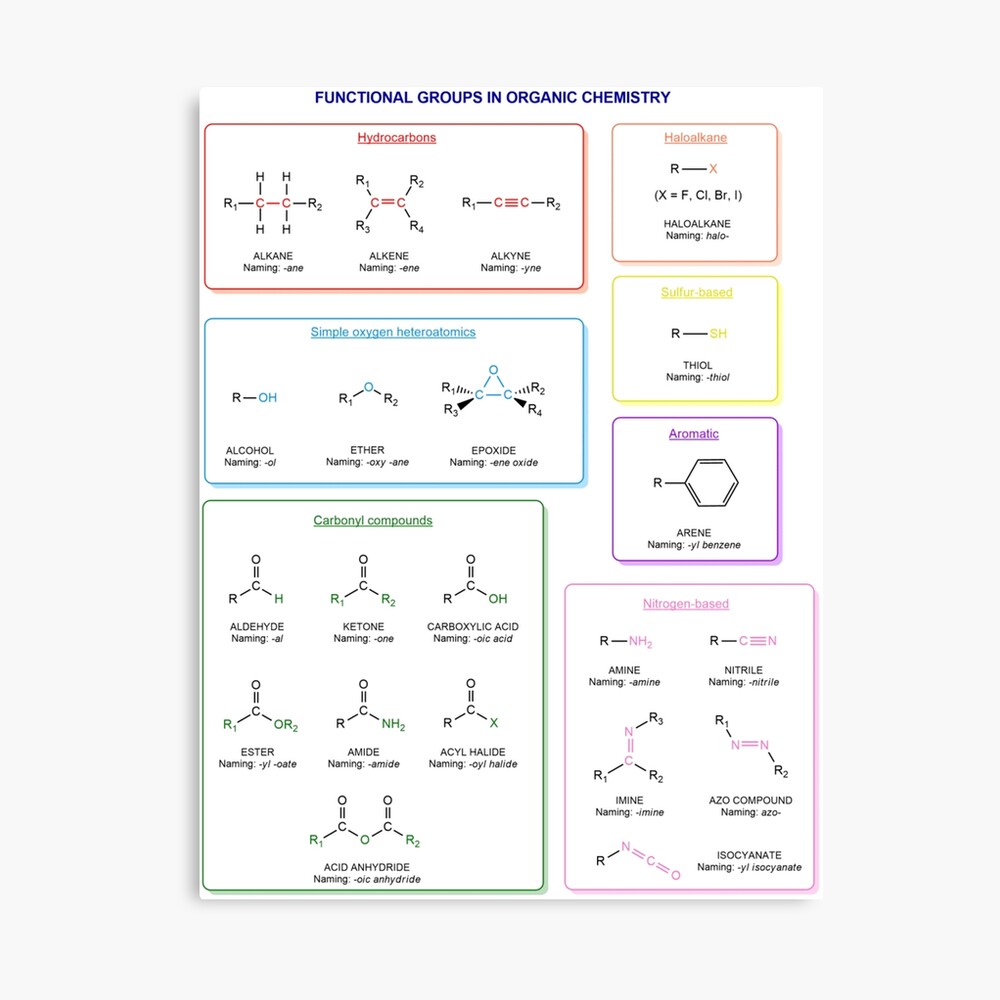
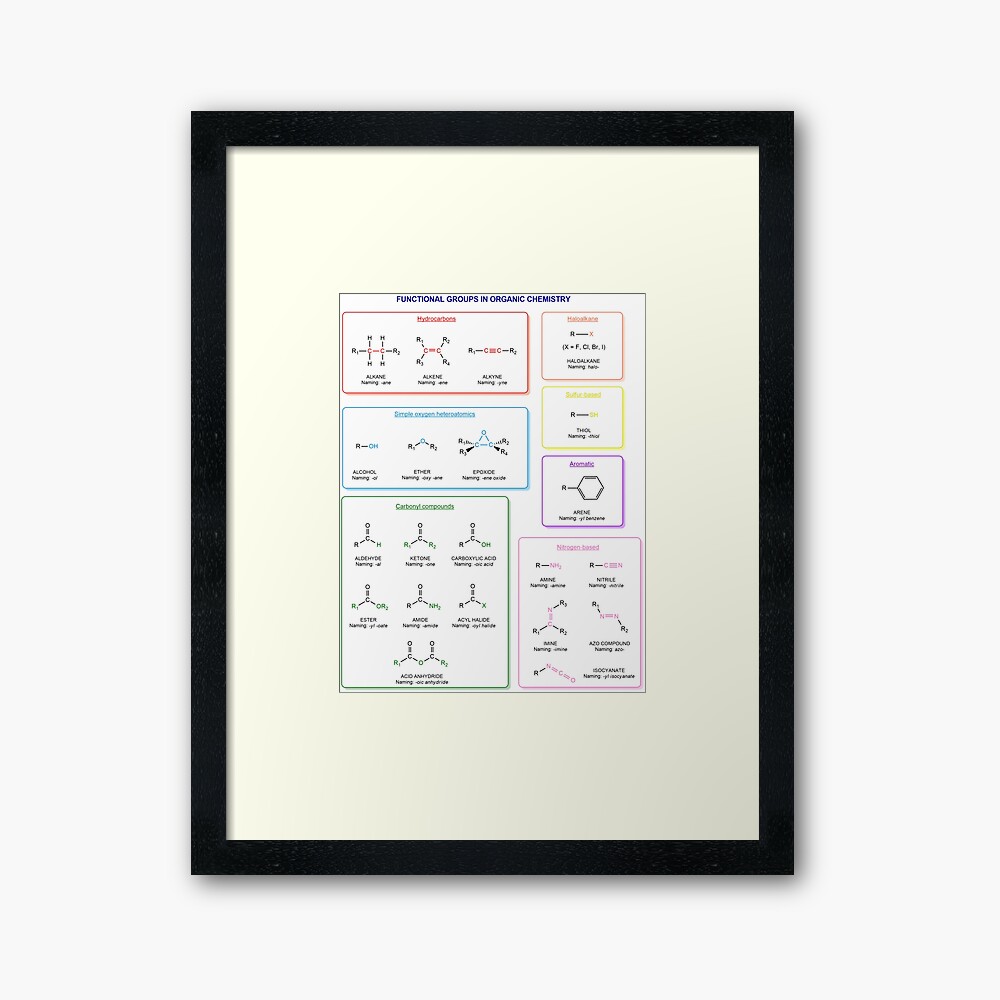
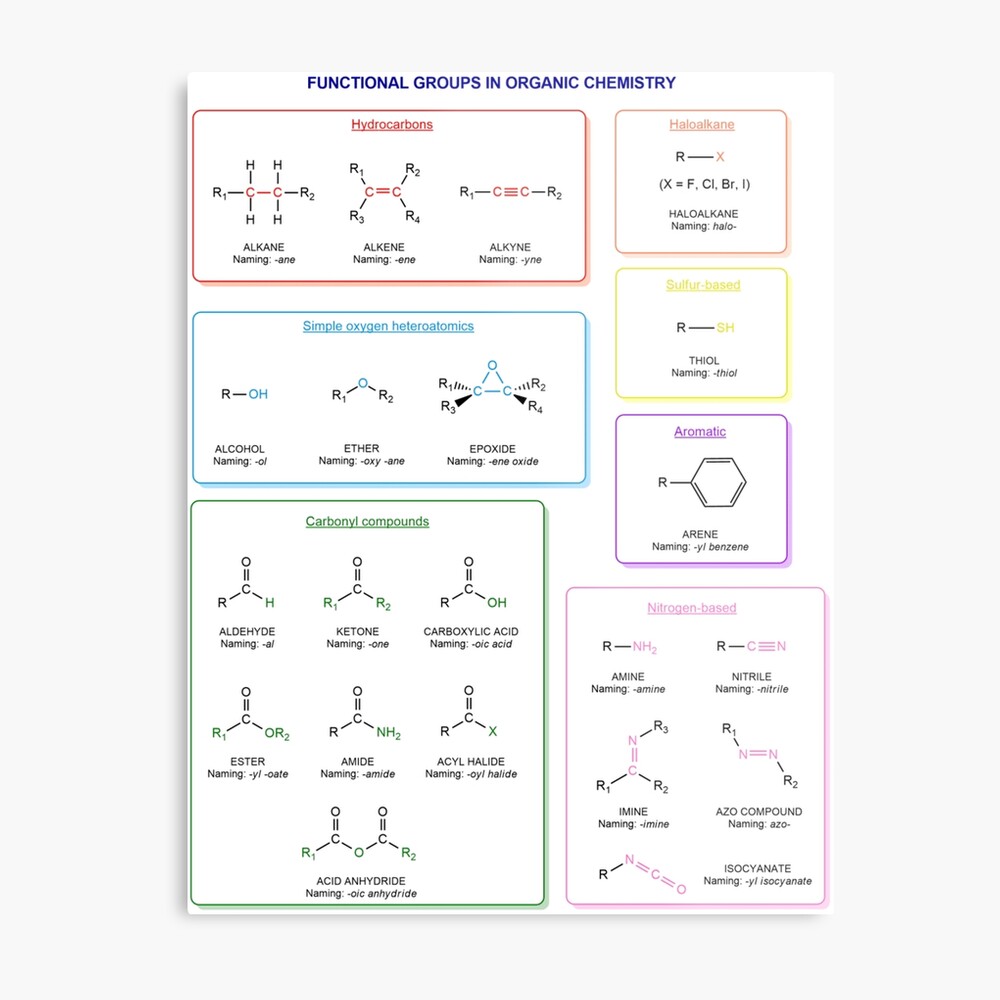
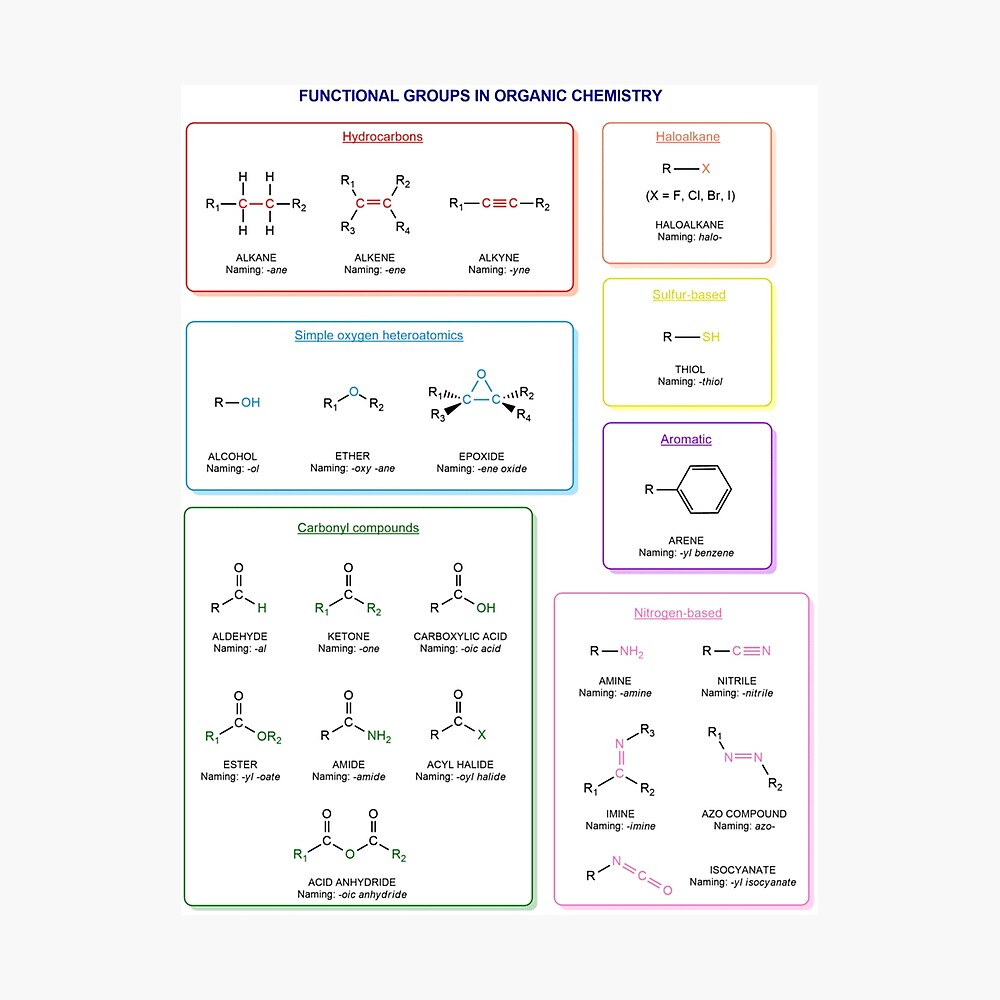

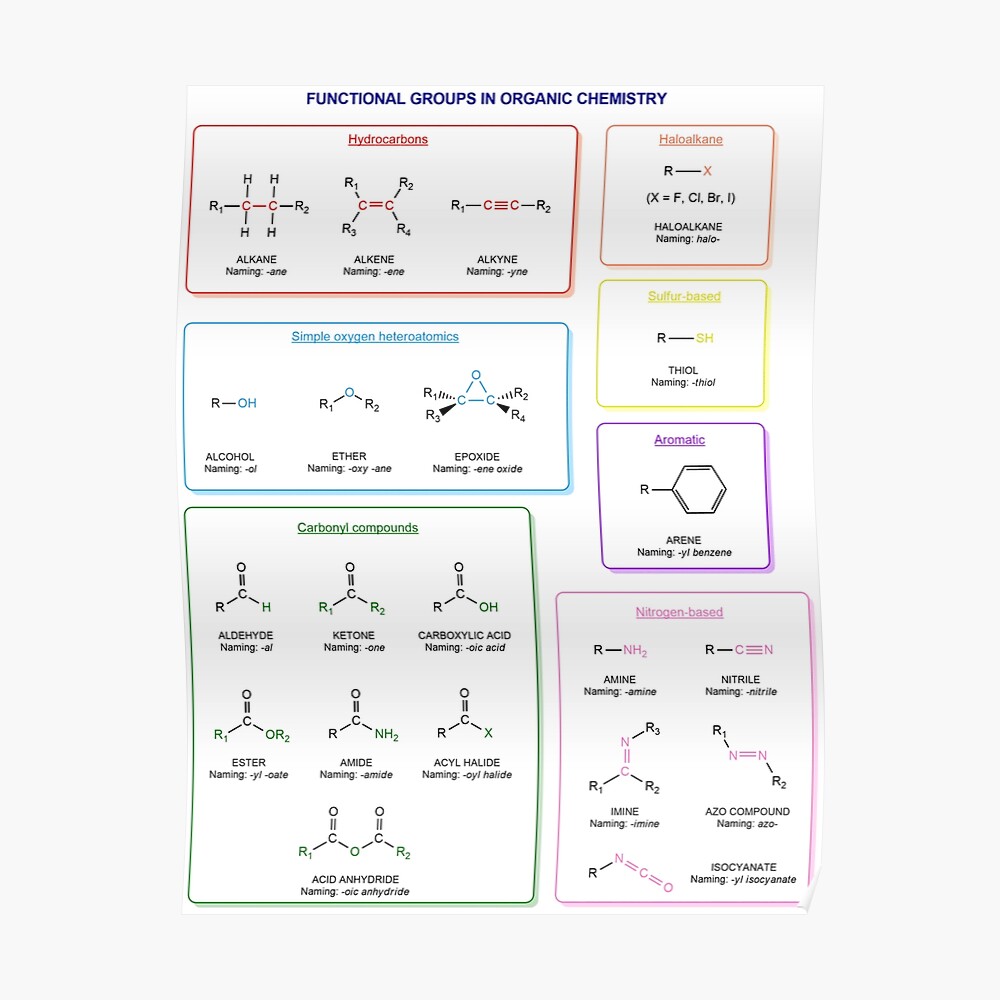
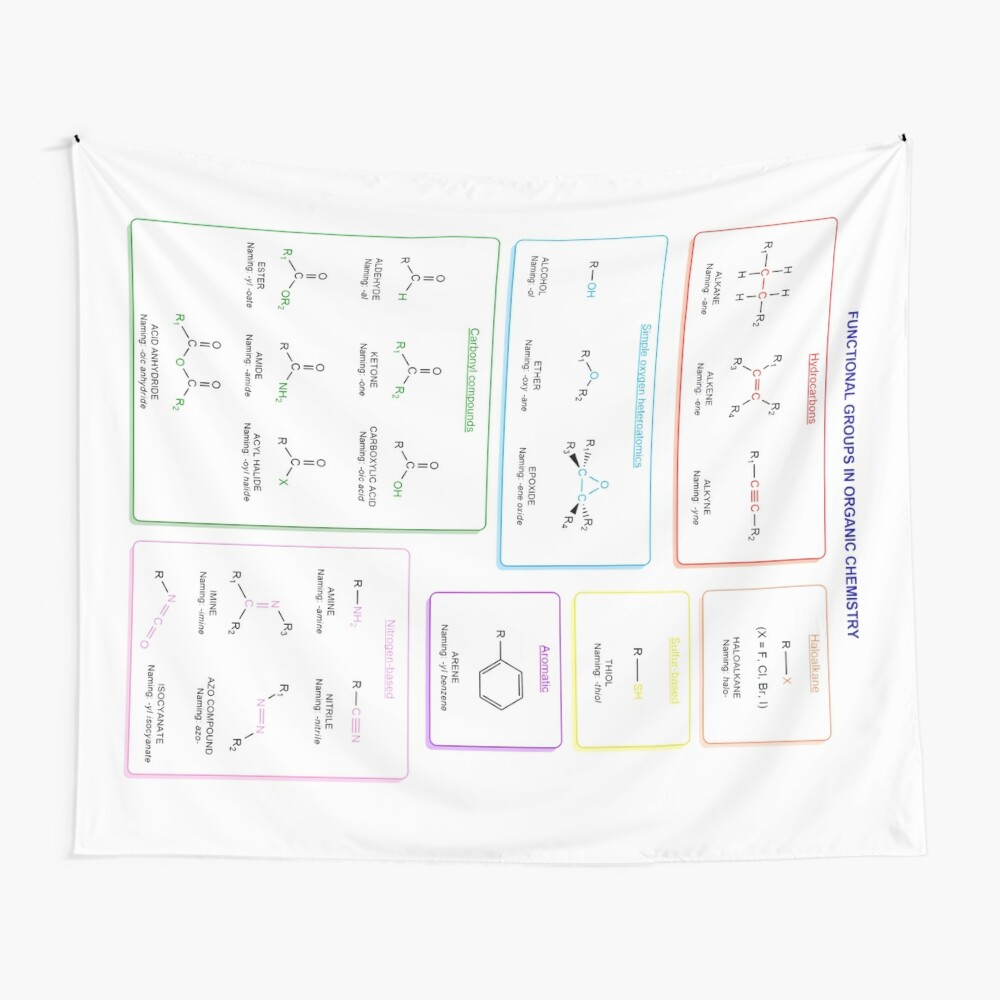


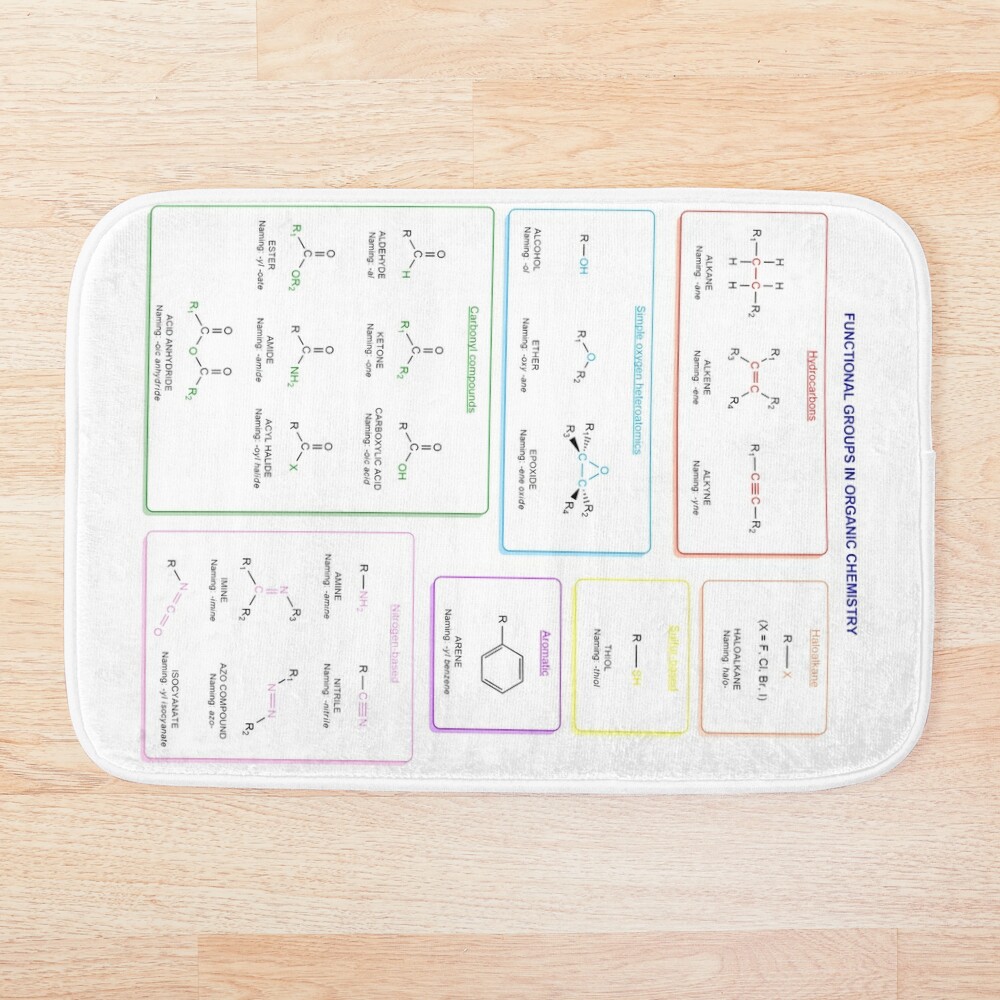


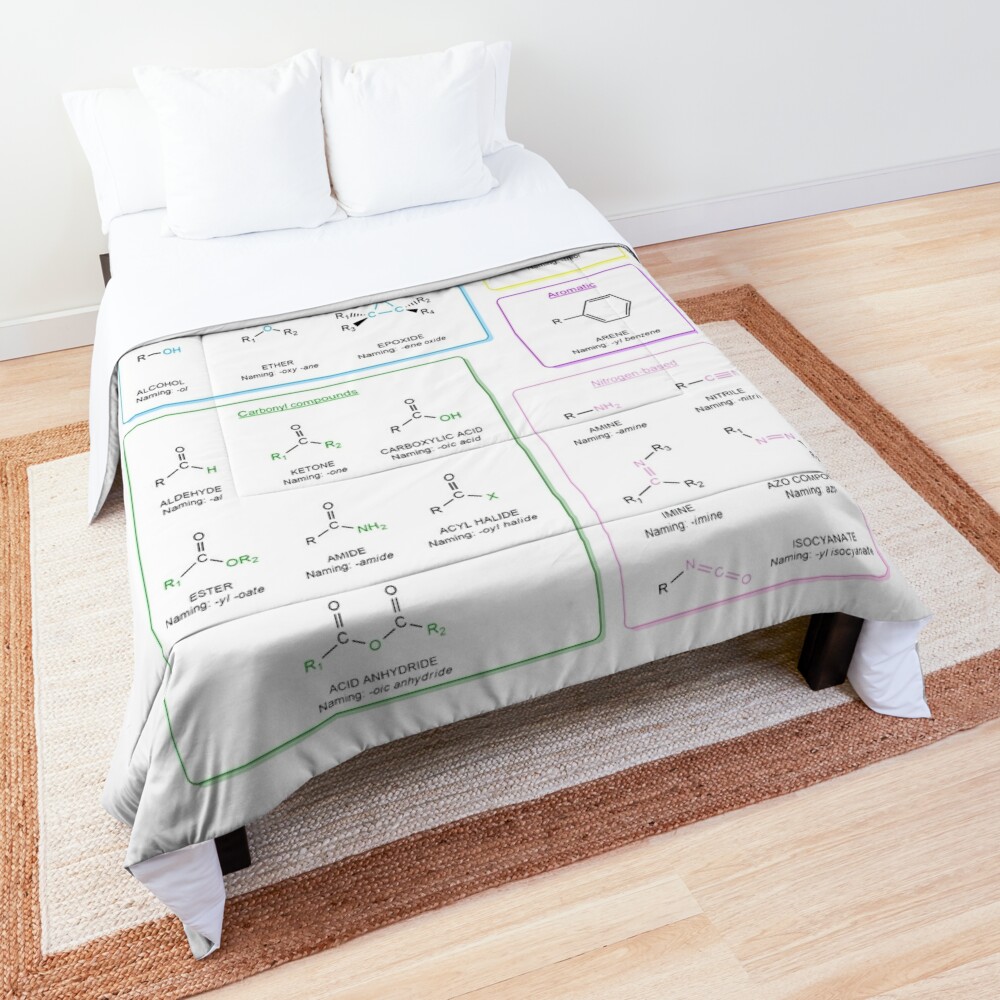
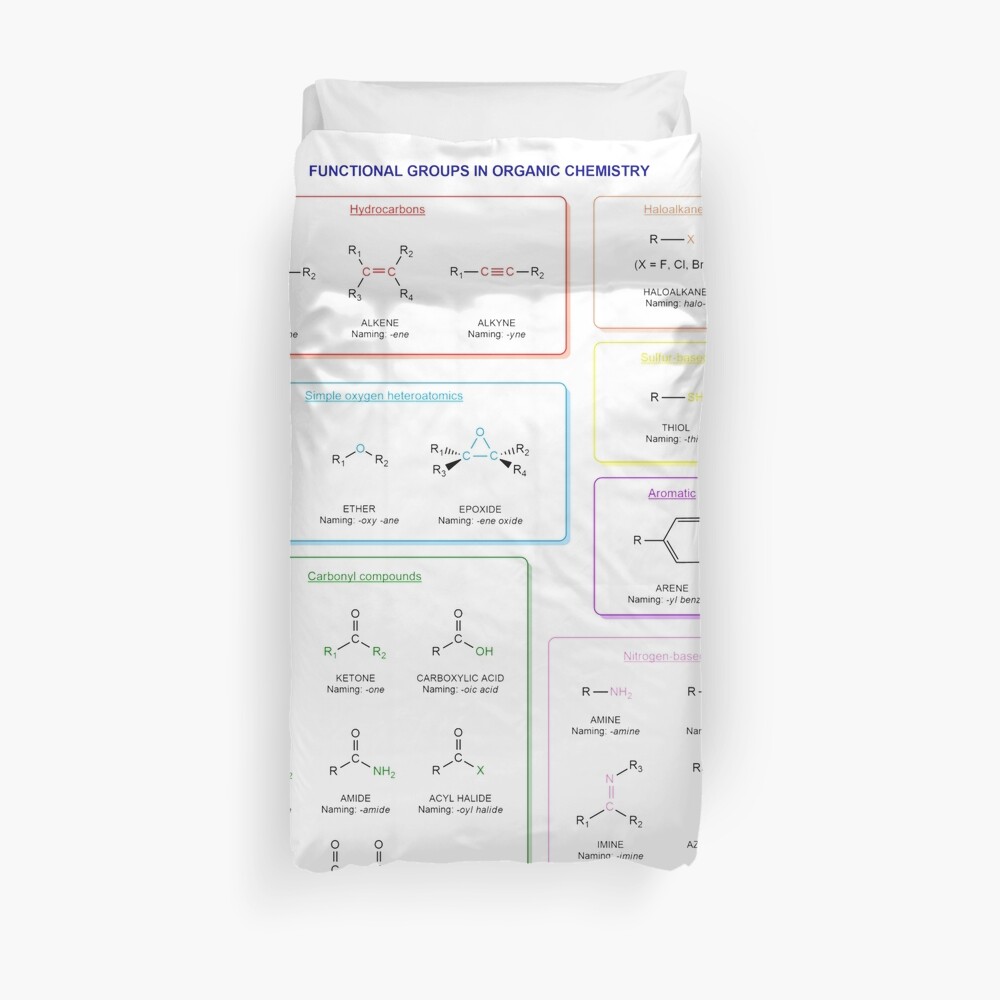
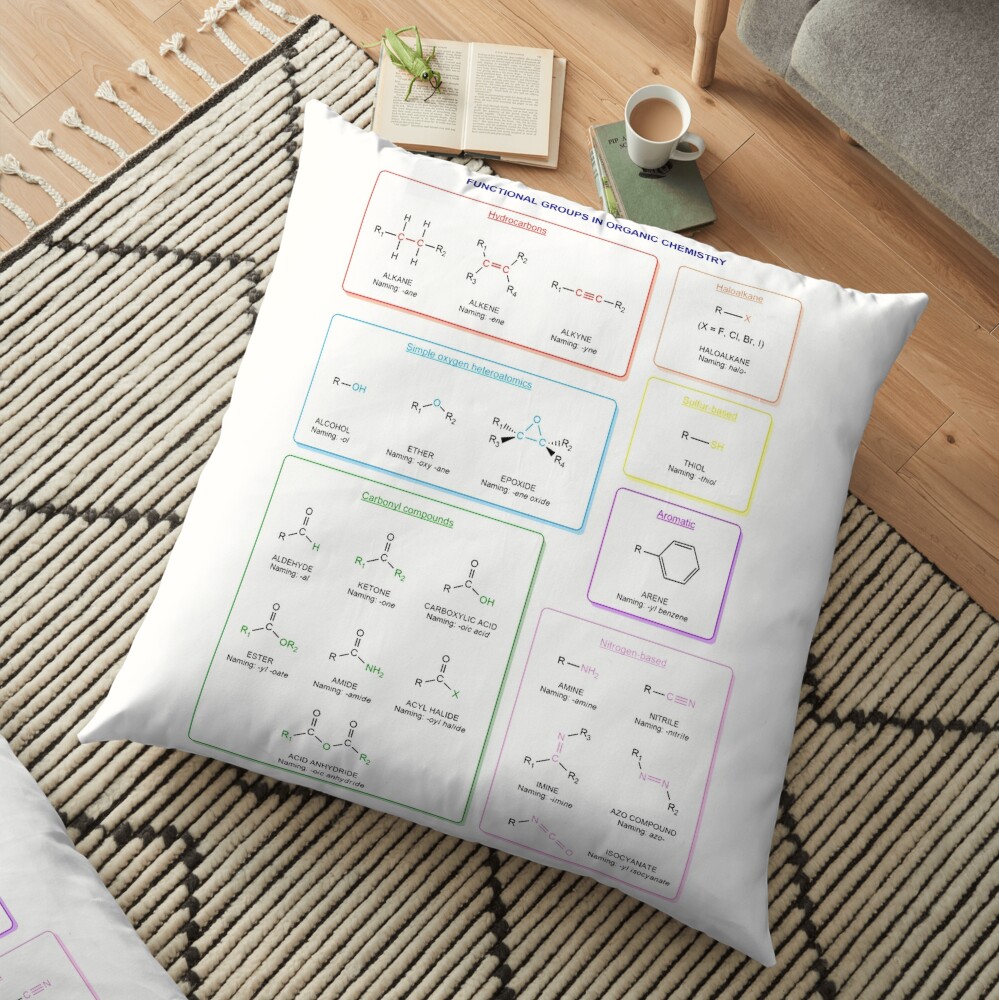


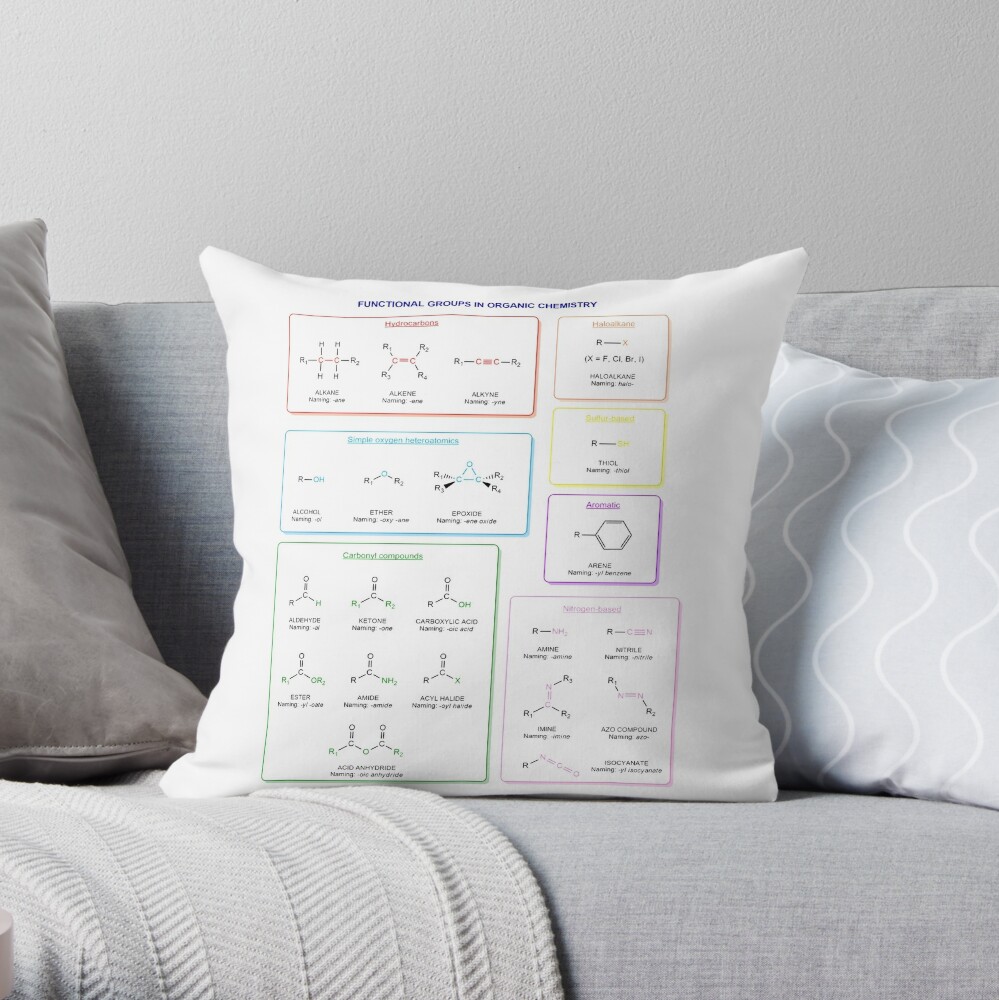


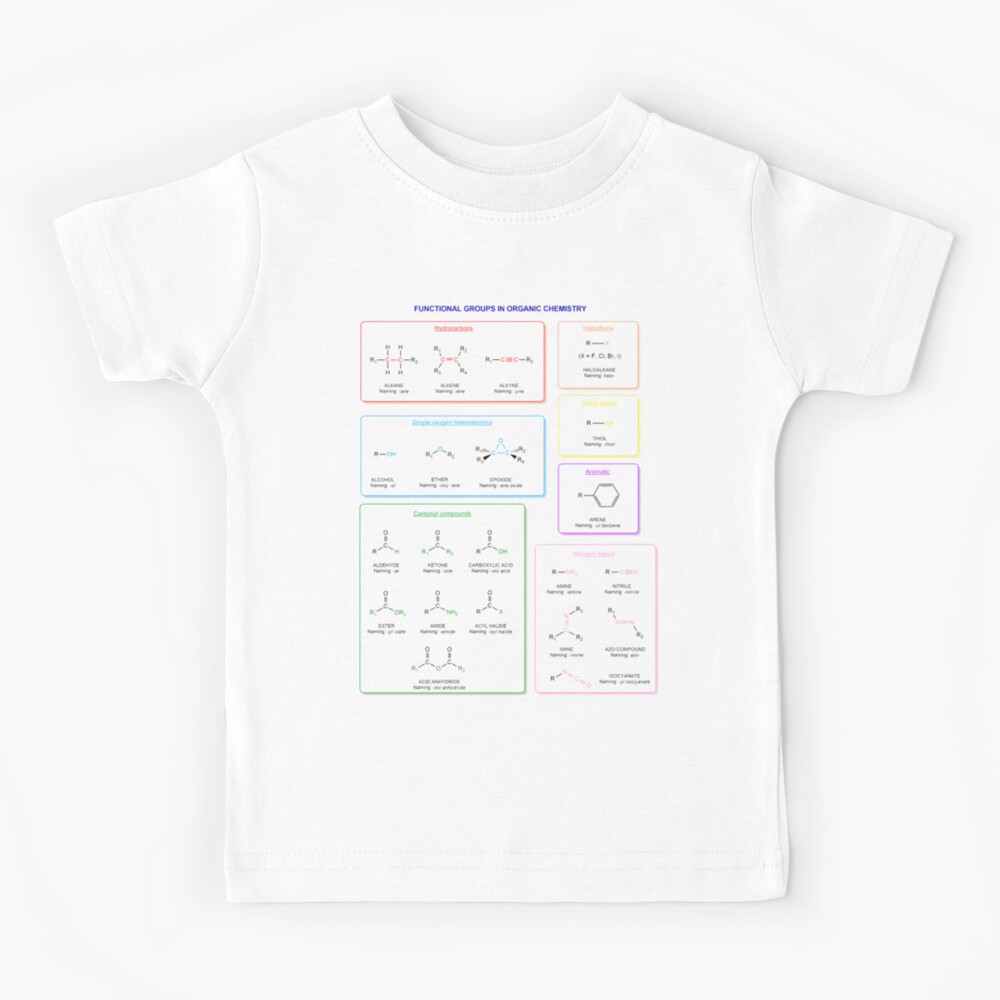



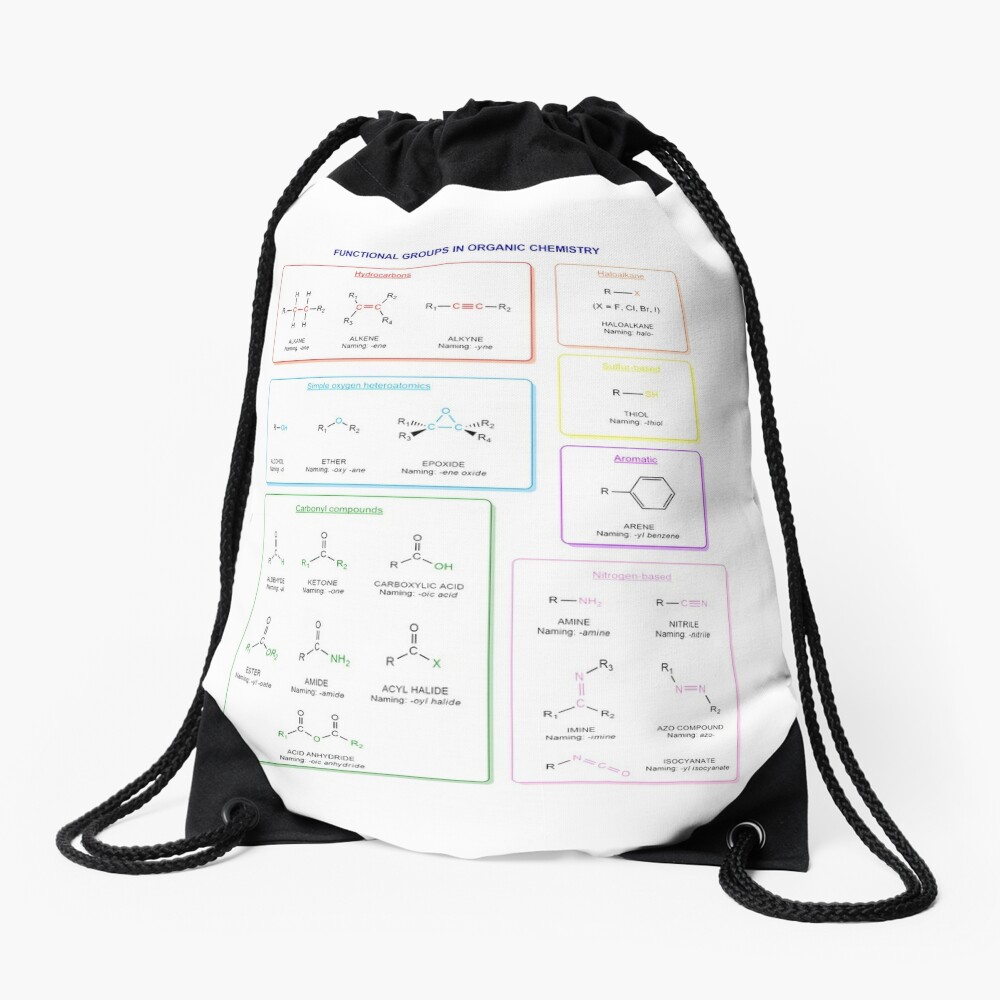



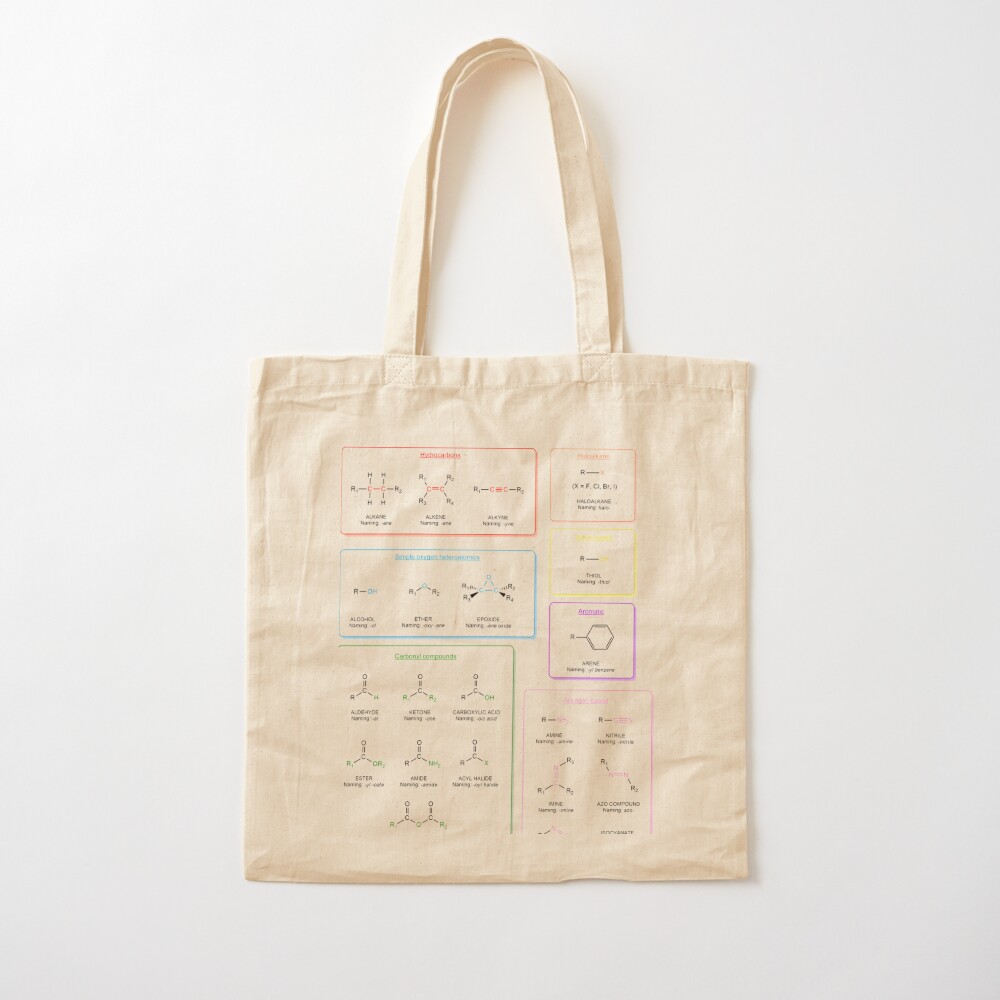


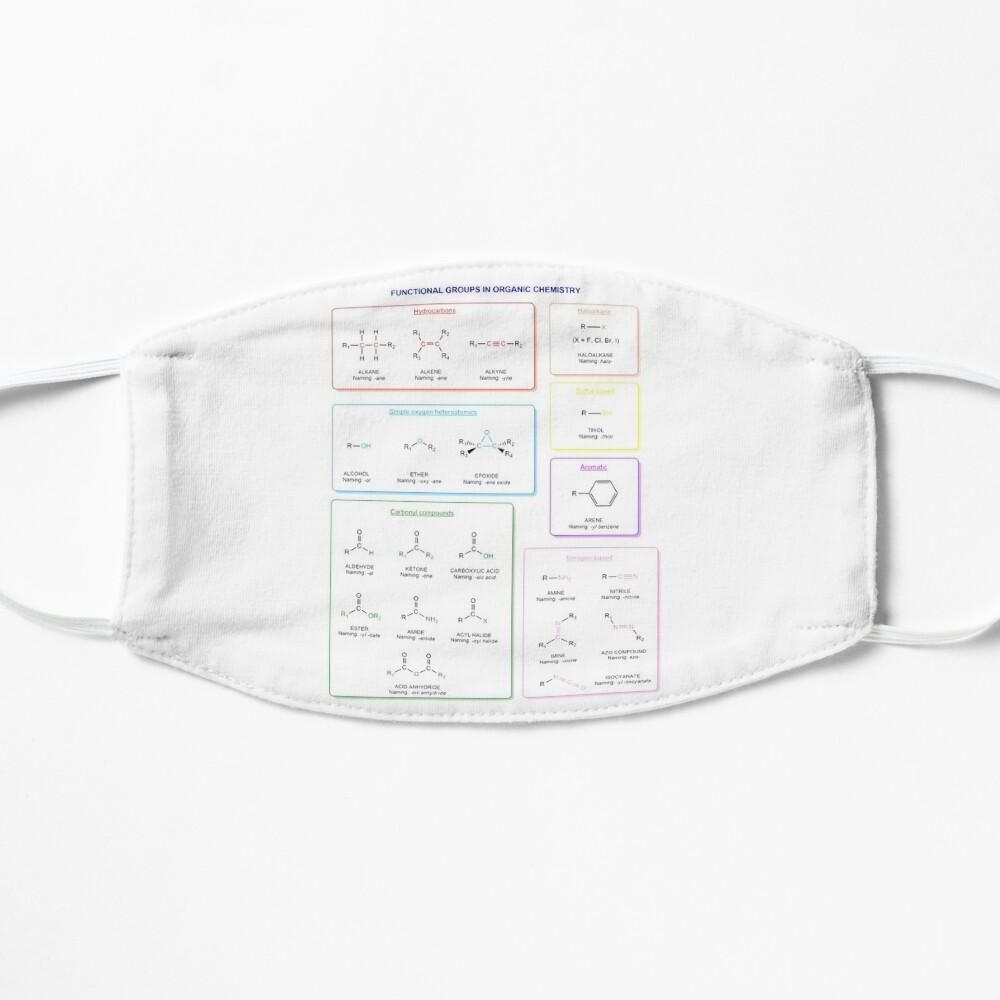

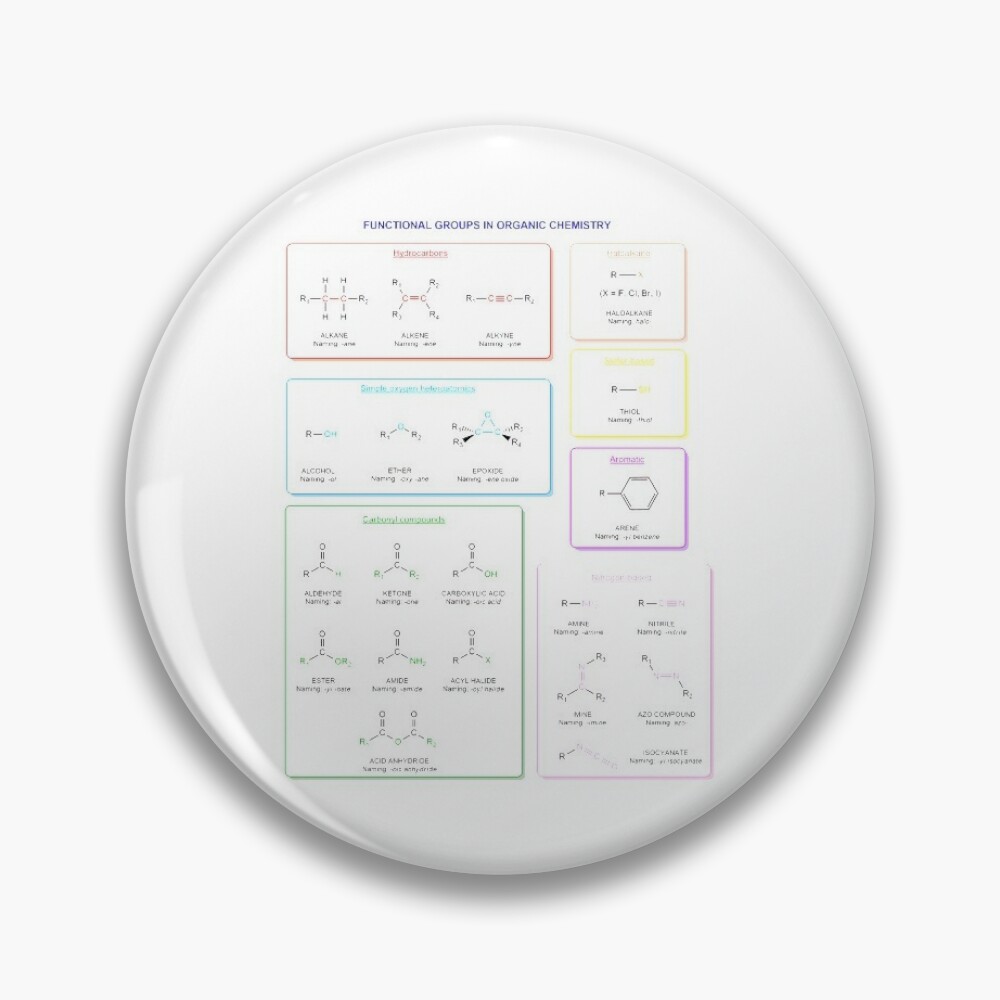
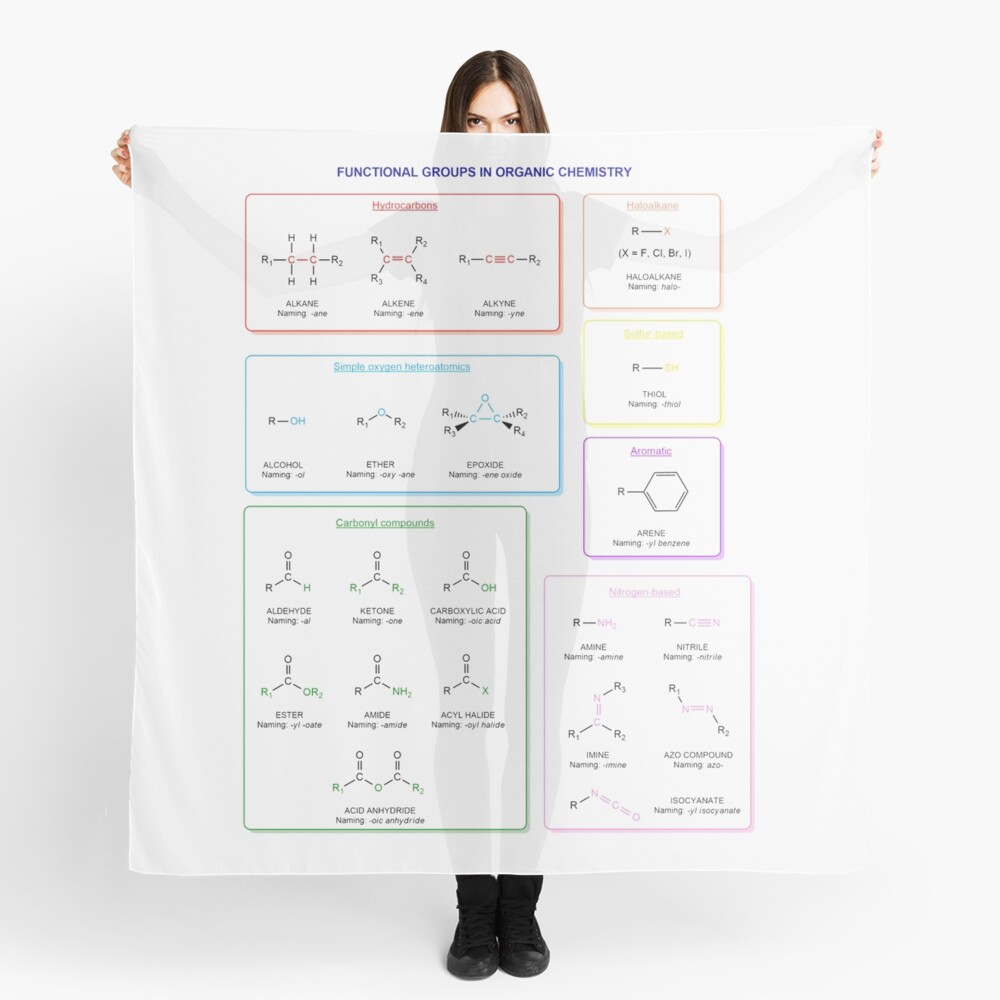









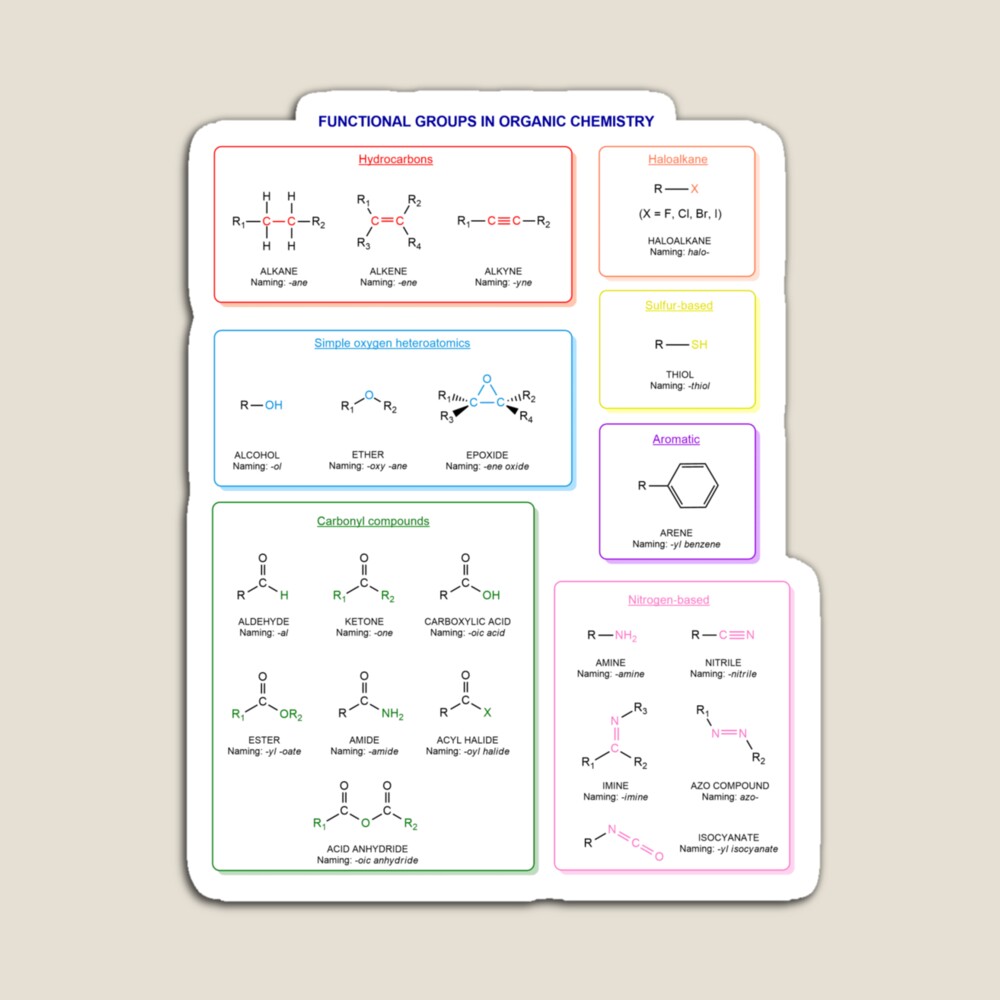
Functional groups in organic chemistry Functional groups are structural features distinguish one organic molecule from another. They determine a molecule’s geometry, physical properties, and reactivity. Actually, a functional group is an atom or a group of atoms with characteristic chemical and physical properties. It is the reactive part of the molecule. The functional group is bonded to the carbon backbone or skeleton which consists of carbon-carbon and carbon-hydrogen σ bonds. These bonds are strong, nonpolar, and not readily broken. This carbon skeleton can contain some other atoms of the so-called heteroatoms. This can be nitrogen, oxygen, sulfur, phosphorus and the halogens. Heteroatoms have lone pairs and create electron-deficient sites on carbon. The carbon skeleton can also contain multiple bonds (π bonds). The most common π bonds occur in carbon-carbon and carbon-oxygen double bonds. They are easily broken in chemical reactions and makes a molecule a base or a nucleophile.
ReplyDelete